Dr. Atul Gawande
About Lifebox Co-founder
Making surgery safer for every patient, every time
Through tools, training, and partnerships, we make surgery and anesthesia safer worldwide.
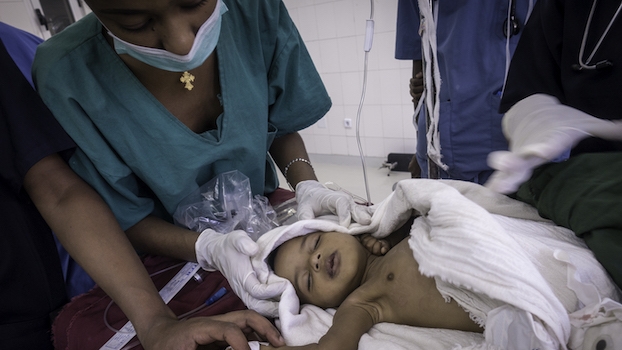
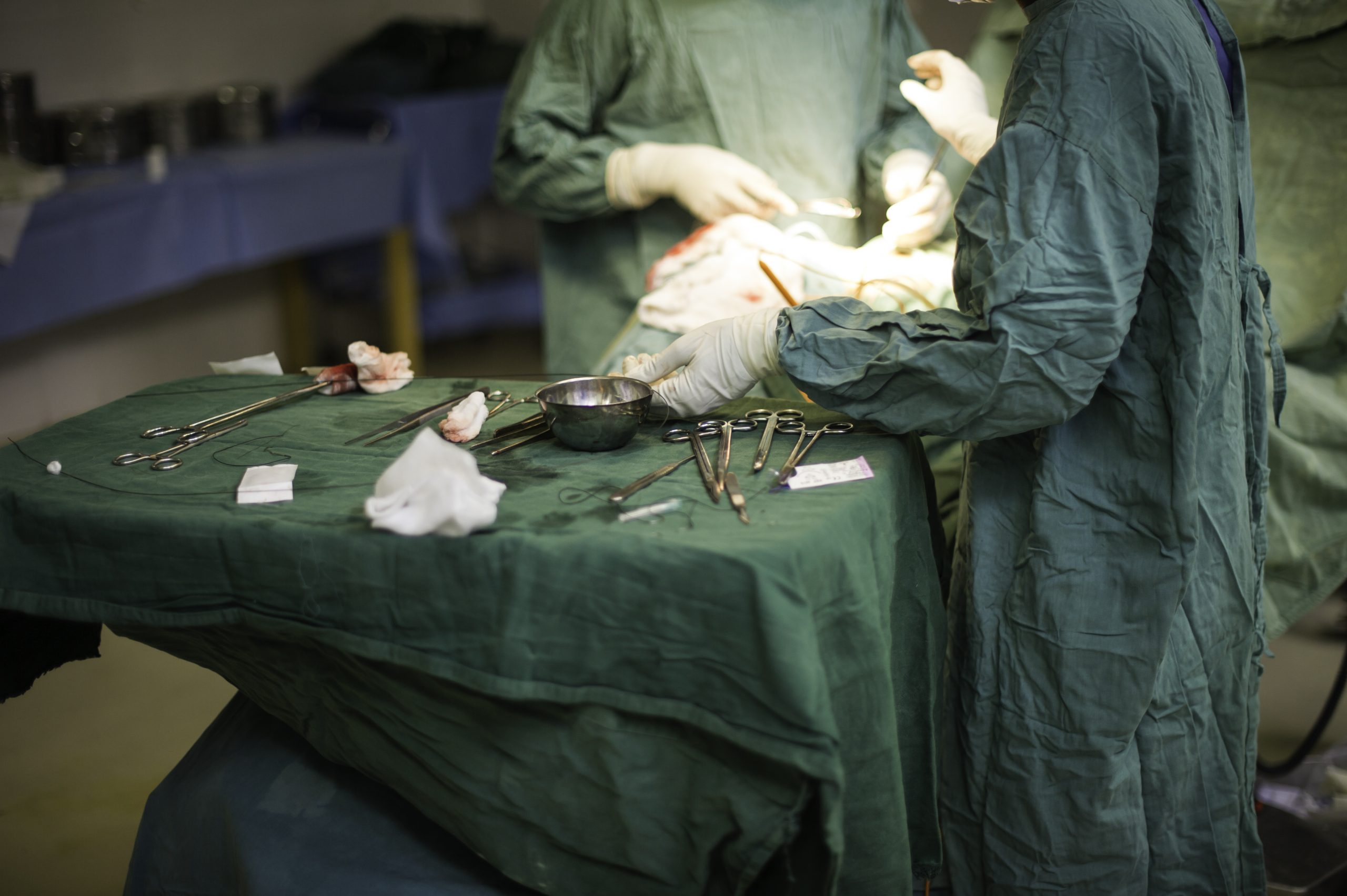
Lifebox was founded by the same individuals that created the WHO Surgical Safety Checklist – including Dr. Atul Gawande. Since 2011, Lifebox has worked in 116 countries, trained more than 12,000 healthcare providers, and made surgery safer for 181 million patients. This work spans improving anesthesia safety – including capnography and pulse oximetry – reducing surgical infection through our Clean Cut program, and strengthening surgical teamwork.
Lifebox is strongly committed to addressing inequities both within our organization and in the global surgery community. We have set ambitious goals for investing in staff, leadership, and innovation in the regions where we work to reduce our footprint in high-income countries. Read more about this work in our strategic plan – which puts multidisciplinary teamwork at the heart of our approach.
For updates on our work, follow Lifebox CEO – Kris Torgeson – on Twitter or sign up for email updates.
“For the first time in history, you’re more likely to die of a surgically treatable condition than an infectious disease. It is critical that we close the gap for the 5 billion people who don’t have access to safe surgery. The magic of Lifebox is that we are creating a movement, making safer surgery possible for patients everywhere and creating a better system as a whole.”Dr. Atul Gawande, Lifebox Co-founder and Immediate Past Chair
The last 10 (plus 1!) years have been absolutely transformative for Lifebox and for the lives we have helped save. In just over a decade, we have
Find out more about our work saving lives through safer surgery.

About Lifebox Co-founder
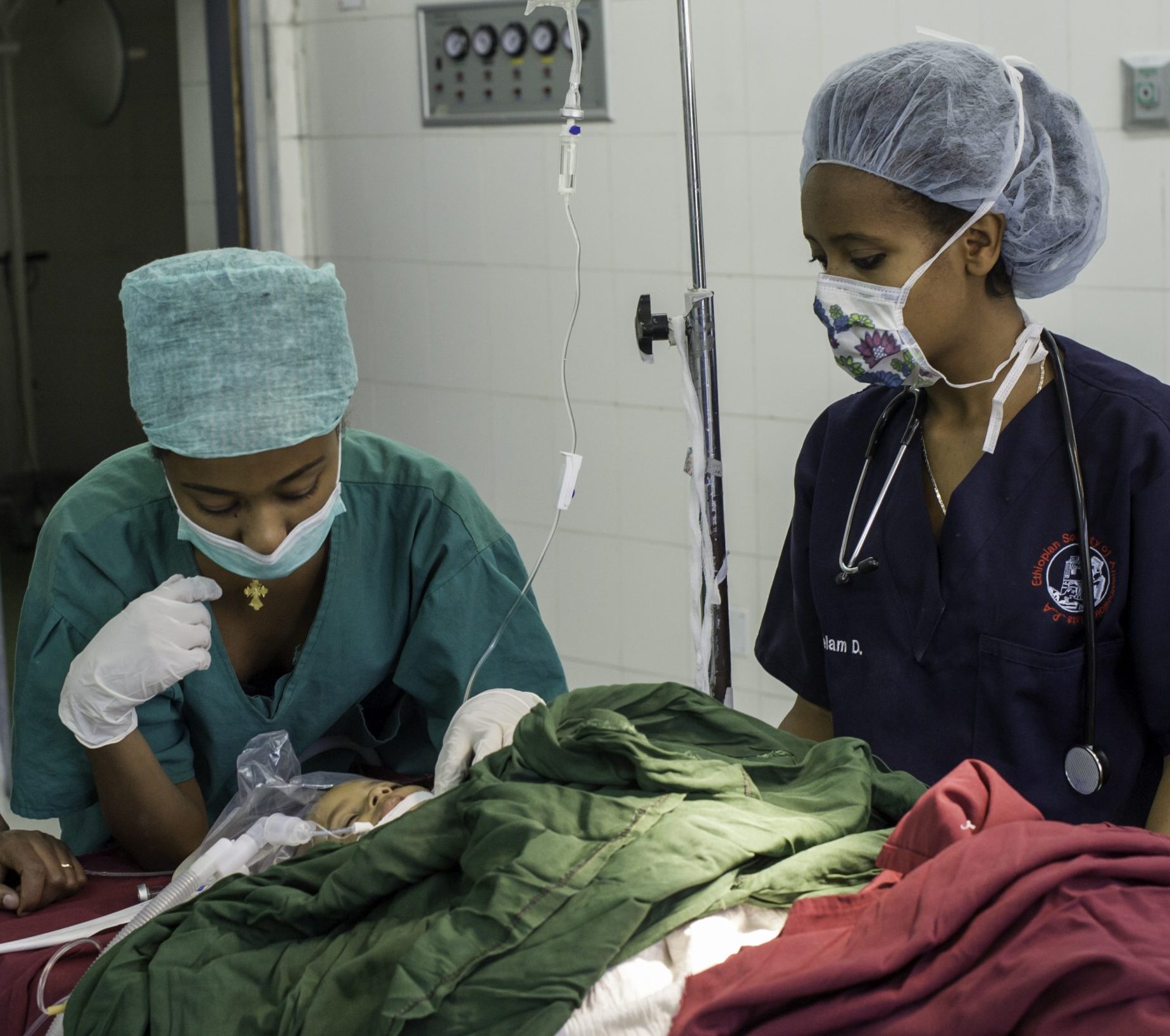
Addressing critical gaps
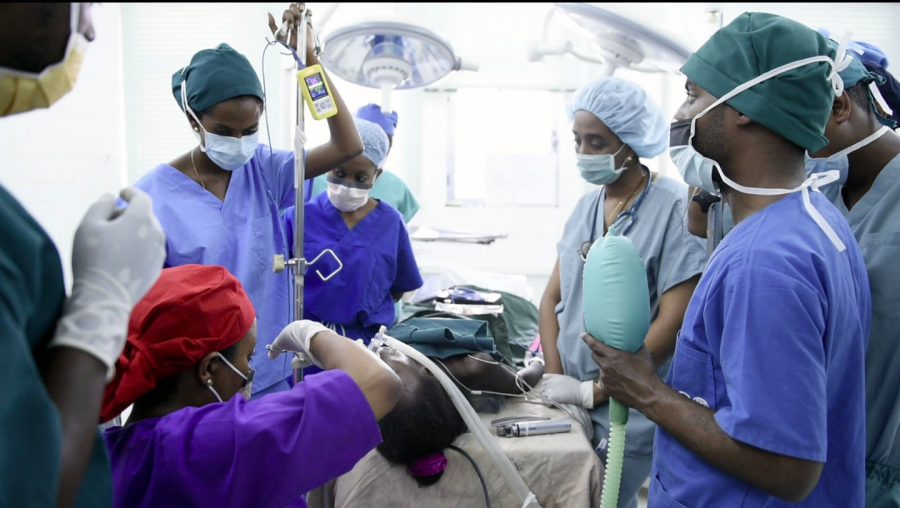
Stronger Teams, Safer Surgery
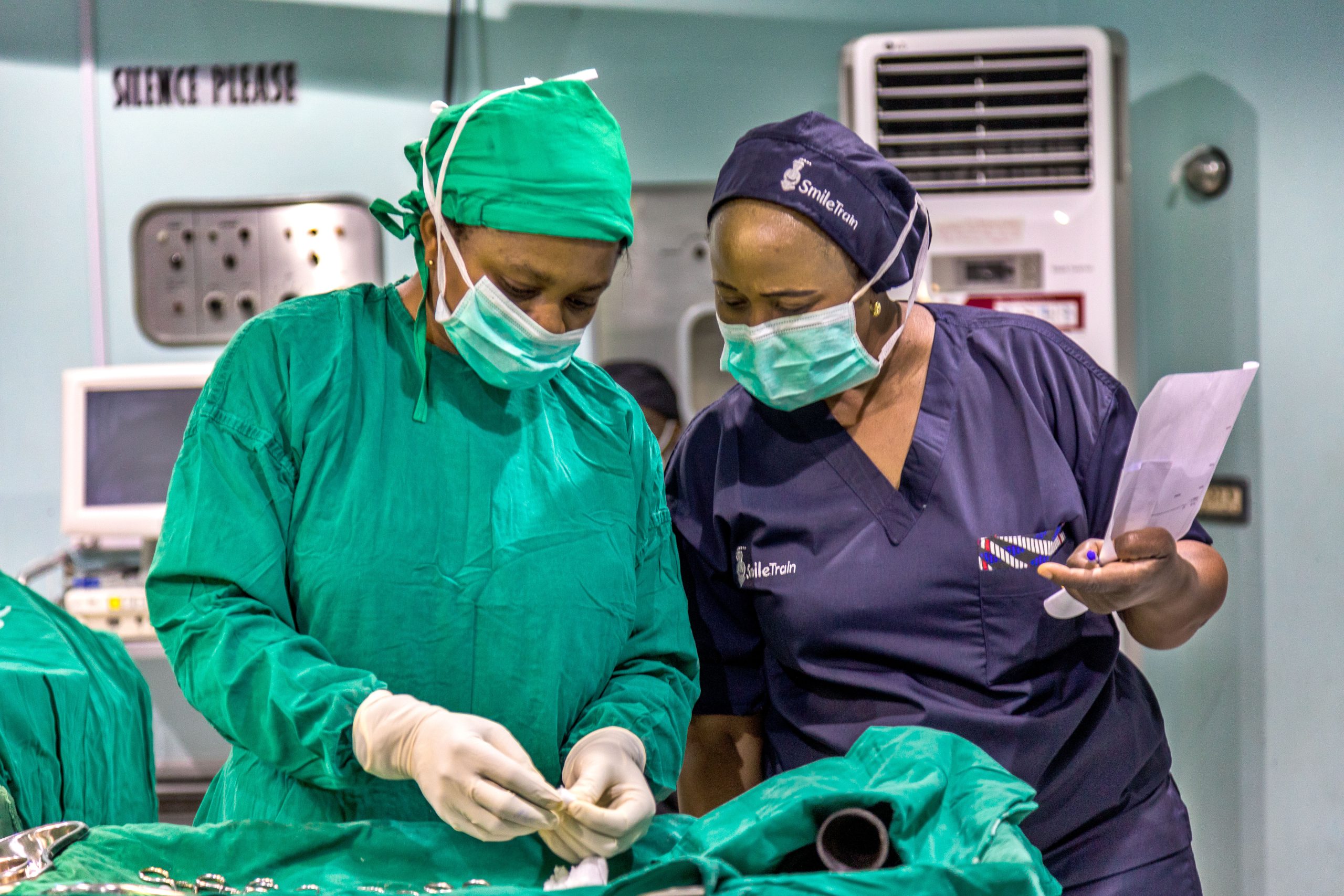
Elevating the quality and safety of cleft and pediatric surgery in more than 70 countries through surgical system strengthening
Lifebox is registered as a charity in England & Wales (1143018), a 501(c)(3) non-profit in the US (EIN 46-2266526) and is a registered nonprofit in Ethiopia. A consortium of partners in Australia and New Zealand – known as Lifebox Australia & New Zealand (ANZ) – raises funds and implements projects.
Lifebox was founded in 2011 by four of the world’s leading medical professional and academic organizations – Association of Anaesthetists, Brigham and Women’s Hospital, Harvard T.H. Chan School of Public Health, and the World Federation of Societies of Anaesthesiologists. Lifebox was launched with the simple aim of providing pulse oximeters to operating rooms that lacked this critical monitoring device.